Mitomycin C (C15H18N4O5) is a chemical product derived from Streptomyces caespitosus, an actinobacteria species. It appears as a blue-violet crystal or powder at room temperature. The product is soluble in water and rapidly degrades in acidic preparations. Healthcare researchers and scientists have studied and applied mitomycin C in cancer treatment for its antitumor properties.

Mitomycin has become a popular chemotherapeutic agent, intravenously administered (intrahepatic) in treatments for upper-gastrointestinal cancers (i.e., stomach and esophageal carcinomas). As the product enters directly via the bloodstream (through a catheter), patients do not experience the usual chemo side-effects such as nausea, alopecia (hair loss), or vomiting. There are currently no pill variants of the mitomycin C drug.
In most cases, healthcare practitioners will prescribe mitomycin C with a combination of other chemotherapy drugs and radiotherapy for improved patient recovery. Mitomycin C, combined with other anticancer agents, may result in elevated serum enzymes during therapy. Healthcare practitioners may also prescribe this compound as a single agent in selective cancer treatments. The side effects of mitomycin C treatment include mouth sores, generalized fatigue, bladder problems, low white and red blood cell counts, and low platelet levels.
Functions and Properties
Medical experts use mitomycin C as an antineoplastic antibiotic, which targets and eliminates rapidly dividing cancer cells. Mitomycin functions as a DNA crosslinking agent, which selectively inhibits cell proliferation and synthesis. The covalent reaction causes a separation of complementary DNA strands, resulting in inhibited replication. Mitomycin C exhibits anti-Gram negative and anti-Gram positive activities and contains three moieties: quinine, urethane, and aziridine groups.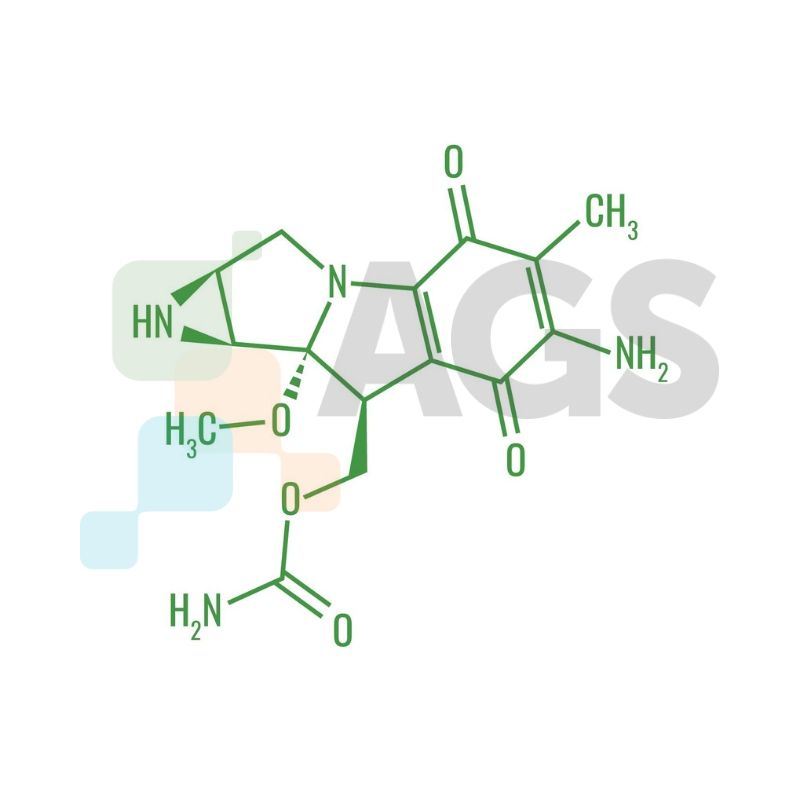
Chemotherapeutic mitomycin drugs usually function with a requirement of enzymatic bioreduction that exerts its biological effects. As such, the alkylation of DNA remains the most popular mechanism for mitomycin C function. However, researchers discovered other mechanisms related to the drug’s effectiveness through redox cycling and RNA inhibition.
One study shows mitomycin targeting thioredoxin reductase in vitro with purified enzymes and cancer cell cultures. This compound can support cancer treatment by disrupting DNA replication within a cell. The process prevents mutated/cancer cells from multiplying, avoiding health complications.
While often intravenously administered, mitomycin may occasionally escape the vascular system, leading to severe cell damage (i.e., the inhibition of protein and RNA production). The complication usually appears as swelling and redness of the skin. Healthcare practitioners require thorough training to administer the drug to prevent complications.
Healthcare Applications
Cancer Treatments
Healthcare specialists apply mitomycin C in various cancer treatments — examples include breast, bladder, lung, pancreas, and stomach cancer treatments. Healthcare providers collect a blood sample from patients to assess the viability of mitomycin treatment before administering mitomycin dosages for chemotherapy. The dosage of mitomycin that patients receive differs according to their height, weight, and health condition.Gastric Cancer
Researchers studied mitomycin C-treated pluripotent stem cells as a delivery method in photothermal therapy for gastric cancers. Specifically, a collaborative study discovered that mitomycin C serves as an effective delivery system for gold nanorods, which generated heat that eliminated human-induced pluripotent stem cells.The research follows a growing interest in stem-cell research as cancer treatment solutions based on the intrinsic tropism toward tumors. High internal pressures in tumors make it a challenge for treatment with conventional anticancer agents (i.e., nanoparticles). In contrast, stem cells with anticancer agents can effectively disseminate within the entire tumor for improved results.
Colorectal Cancer
Researchers discovered that Pegylated liposomal (PL) and mitomycin C lipidic prodrug (MLP) suggest an effective treatment for patients suffering from metastasized colorectal carcinoma. Scientists based the findings on a phase 1A/B study of the PL-MLP drug combination. The study results showed 42 percent of stable disease in efficacy-valuable patients, along with longer survival, suggesting the improved probability of disease control.Lung Cancer
Twenty-five percent of lung tumors involve activated mutations in the KEAP1-NRF2 pathway, which result in aggressive tumor growth and poor patient prognosis. While there are currently no approved drugs in targeting or resolving mutations in the KEAP1-NRF2 pathway, researchers have identified mitomycin C as a potential treatment solution against NRF2 for its high cytotoxic values.Bladder Cancer
Scientists discovered that electromotive drug administration (EMDA) of mitomycin C combined with BCG (Bacillus Calmette–Guerin) provides a viable alternative for invasive nonmuscle bladder patients unresponsive to BCG therapy. Results from the study showed a 50 percent progression-free survival rate in patients.Recent research established the pressing need to solubilize and stabilize mitomycin C solutions for intravesical (occurring within the bladder) administration, in response to the concerns of inferior drug delivery. The study involved mitomycin C solutions prepared with conventional and alternative compounding methods. Researchers determined the quality of pharmacologically prepared mitomycin C solutions by studying various factors involving physical and chemical stability. Subsequently, the experiment included incubation of solutions at 50 °C for 50 minutes followed by storage at 37 °C, sufficient to solubilize the drug. Chemical degradation of MMC solutions was determined over six hours using high-performance liquid chromatography (HPLC) analytics, while checking for physical stability in parallel.
Results from the study suggested that conventional admixture preparations of mitomycin C may lead to compromised solubility, which decreases drug exposure within the bladder and reduces treatment effectiveness. While alternative compounding methods may increase drug exposure with improved solubility, medical providers need to monitor patient progress for additional mitomycin toxicities that may lead to health complications. The experiment illustrates the importance in balancing preparation and patient safety for optimal results in applications of this antibiotic.
Breast Cancer
This compound has been actively applied in advanced breast cancer treatments through combined chemotherapy with a series of other drugs such as gemcitabine, vinblastine, vindesine, and vinorelbine. Researchers also discovered that mitomycin C and methotrexate combination chemotherapies proved effective against metastatic breast cancer following aggressive treatment with anthracycline, taxane, capecitabine, and vinorelbine (ATCV). The applications of mitomycin C expand beyond cancer treatments, with multiple use cases across the medical field.Treatment of Persistent Acinetobacter baumannii
Acinetobacter baumannii is an opportunistic, rod-shaped, Gram-negative type of bacteria known for causing infections in healthcare settings. Additionally, A. baumannii exhibits intrinsic antibiotic-resistant properties, which complicates patient treatment.
Researchers repurposed four anticancer drugs to inhibit A. baumannii growth, with mitomycin exhibiting the most success. The study showed that mitomycin C was the most effective drug, with the highest cell growth inhibition rate, affecting stationary-phase, persister, and biofilm cells. The test results drew a similarity between tumor growth and bacterial infection and how mitomycin’s cytotoxic properties can eliminate A. baumannii populations.
Therapeutic Strategy for Parkinson’s Disease
Mitomycin C may assist in the treatment of Parkinson’s disease. Researchers experimented with mitomycin-treated undifferentiated embryonic stem cells in a mouse model. The treatment of mouse embryonic stem cell cultures resulted in reduced tumor formations and improved brain function over a 15-month period.The research discovered that test subjects experienced significant motor function improvements and reduced akinesia (the loss of voluntary muscle movements), as observed in people with Parkinson’s. Additionally, by halting mitotic function (chromosome replication) in undifferentiated embryonic stem cells, the treatment resulted in subjects exhibiting a marked improvement in dopamine release (linked to increased alertness and improved mood).
Ophthalmic Uses — Slowing Down Fibroblast Formation in Glaucoma
Glaucoma is an eye condition that causes gradual damage to the optic nerve and remains a significant cause of blindness in individuals aged 60 and above. Health care specialists commonly apply mitomycin C in glaucoma surgery (i.e., phacoemulsification and trabeculectomy procedures), which reduces scarring by inhibiting the function of scar tissue cells.
Specifically, mitomycin inhibits fibroblast proliferation and microvascular blood supply, which otherwise lead to scarring that stops aqueous outflow during a surgical procedure. Healthcare practitioners may physically apply mitomycin C solutions on the site of surgery using saturated sponges. Mitomycin C treatment options serve as practical solutions for patients who are unresponsive or sensitive to laser and other medical treatments. Similarly, specialists have applied mitomycin C in preventing haze build-ups after PRK or LASIK surgery. Eye specialists have also used mitomycin C to treat strabismus (the misalignment of the eyes when looking at an object), which reportedly reduces the number of post-surgery adhesions (bands of scar-like tissue).
Managing Caustic Esophageal Stricture
Esophageal strictures occur with the narrowing of the esophagus. Strictures usually occur due to chronic conditions such as GERD, where the structure experiences prolonged exposure to gastric reflux, leading to inflammation, scarring, and narrowing. Caustic esophageal strictures occur due to accidental ingestion of caustic substances (such as batteries) and usually involve children from developing countries. Researchers discovered that mitomycin C provides effective management of caustic esophageal strictures.
The double-blind study reports that topical mitomycin supports post-endoscopic dilation procedures required in esophageal stricture treatment. Results showed that 80 percent of strictures prescribed with mitomycin C treatment exhibited complete recovery compared to 35 percent in the placebo group. As such, mitomycin C may serve as an effective treatment for dysphagia in esophageal stricture cases. Mitomycin C remains an integral research component in academic and lab-based research, specifically as a prototype of alkylating anticancer treatments. Unlike 5-FU treatments, mitomycin treatment does not rely on cell cycle phases, and provides consistent reactions and results for quality research needs.
Your Ally in Discovery
AG Scientific is a trusted supplier of quality scientific components for academic and research purposes. Our products undergo the strictest quality checks to ensure that customers receive the best products. Browse our comprehensive inventory, including mitomycin C and mitomycin C in NaCl preparations, and find out how we can become your ally in discovery.
Additional Reading
-
Avoiding Cross-Contamination in Antibiotic Manufacturing
-
Single-Use Biosensors for the Detection of Biomarkers